
Concept explainers
Interpretation: To write an equation for the conversion of pyruvate to lactate in the Cori cycle that includes the structures of lactate and pyruvate.
Concept introduction: Glucose is converted to pyruvate by glycolysis
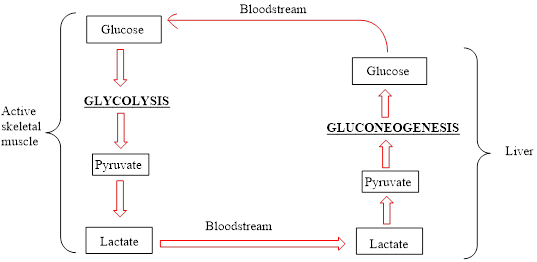
The Cori cycle is named after its discoverers, Gerty Radnitz Cori, and Carl Cori.
An overview of the Cori cycle is as follows:
Want to see the full answer?
Check out a sample textbook solution
Chapter 24 Solutions
EBK GENERAL, ORGANIC, AND BIOLOGICAL CH
- The glucose/glucose-6-phosphate substrate cycle involves distinct reactions of glycolysis and gluconcogenesis that interconvert these two metabolites. Assume that under physiological conditions, [ATP] = [ADP] and [Pi] =1 mM. Consider the following glycolytic reaction catalyzed by hexokinase: ATP + glucose = AG' = -16.7 kJ/mol ADP + glucose-6-phosphate (a) Calculate the equilibrium constant (K) for this reaction at 298 K, and from that, calculate the maximum [glucose-6-phosphate]/[glucose] ratio that would exist under conditions where the reaction is still thermody- namically favorable. (b) The reverse of this interconversion in gluconeogenesis is catalyzed by glucose-6-phosphatase: glucose-6-phosphate + H,0 = glucose + P, AGr = -13.8 kJ/mol K= 262 for this reaction. Calculate the maximum ratio of [glucose]/ [glucose-6-phosphate] that would exist under conditions where the reaction is still thermodynamically favorable. (c) Under what cellular conditions would both directions in the…arrow_forwardLactic acidosis, weakness, fatigue, and sleepiness are observed in patients with genetic defects of one if the enzymes of pyruvate dehydrogenase complex. Explain The causes of the elevation of lactate in the blood of such patients and the emergence of hypoenergetic state. For this purpose write the following: a) scheme of the metabolic pathway of glucose metabolism, that includes this reaction and calculates the energetic effect of the process, outlining the reactions coupled with the use and formation of ATP. b) reaction of the reduction of pyruvate and explain the reason for its acceleration inpatients that have such genetic defects.arrow_forwardThe reactions of the pyruvate dehydrogenase complex are required to generate the substrate that is fed into the TCA (Kreb’s) cycle from pyruvate. The 3 enzymes that make up this complex are pyruvate dehydrogenase (E1), dihydrolipoyl transacetylase (E2) dihydrolipoyl dehydrogenase (E3). a. Name the one diffusible reaction product (i.e. the product that is free to leave the enzyme complex) of each enzyme of the complex. b. Draw the “business end” of the fully reduced form of lipoic acid. c. Using words, fully describe the function of E3 in this complex. Your answer should include all cofactors used, all intermediates and products of this enzyme. DO NOT show any mechanisms for this part.arrow_forward
- The conversion of 1 mol of fructose 1,6-bisphosphate to 2 mol of pyruvate by the glycolytic pathway results in what type of net formation?arrow_forwardWrite a balanced equation for the synthesis of PRPP from glucose through the oxidative branch of the pentose phosphate pathway.arrow_forward[1,6-(C-14)-2,5-(C-13)]glucose Trace the course of through glycolysis and the TCA cycle. You need only to illustrate the structures of D-glucose, labeling the atoms, and of pyruvate, labeling the origin of the atoms in glucose and the distribution of the carbon isotopes in pyruvate. After one round of the TCA cycle where will the labeled carbons be found?arrow_forward
- If the pyruvate is labelled with 14C at its carbonyl carbon, how many rounds of the TCA cycle are required before 14CO2 is released?arrow_forwardThe glucose/glucose-6-phosphate substrate cycle involves distinct reactions of glycolysis and gluconeogenesis that interconvert these two metabolites. Assume that under physiological conditions, [ATP] = [ADP]; [P;] = 1 mM. Consider the glycolytic reaction catalyzed by hexokinase: ATP + glucose ADP + glucose-6-phosphate AG = - 16.7 kJ/mol (a) Calculate the equilibrium constant (K) for this reaction at 298°K, and from that, calculate the maximum [glucose-6-phosphate]/ Iglucose] ratio that would exist under conditions where the reaction is still thermodynamically favorable. (b) Reversal of this interconversion in gluconeogenesis is catalyzed by glucose-6-phosphatase: glucose-6-phosphate + H20 = glucose + P AG" = -13.8 kJ/molarrow_forwardWhich one of the five steps of the pyruvate dehydrogenase complex reaction is most likely to be metabolically irreversible? Explain.arrow_forward
- In what condition and why is pyruvate converted to lactate?arrow_forwardDuring gluconeogenesis, the three irreversible steps of glycolysis have to be bypassed. The first step is the conversion of pyruvate to phosphoenolpyruvate. Which of the following statement is false regarding the reaction step? Select one: a. Conversion of oxaloacetate from pyruvate occurs in mitochondria and shuttled into the cytosol. b. Formation of phosphoenolpyruvate requires both ATP and GTP as an energy source. c. Acetyl CoA is an activator of enzyme pyruvate carboxylase. d. This reaction involves two-step process catalyzed by pyruvate carboxylase and phosphoenolpyruvate carboxykinasearrow_forwardIn the citric acid cycle, malate dehydrogenase catalyzes the following reaction: Malate + NAD+ à oxaloacetate + NADH Calculate the standard free energy change for this reaction (∆Go’). Will it proceed as written under standard conditions? What do you think affects the actual free energy change (∆G) allowing for formation of oxaloacetate from malate?arrow_forward
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education





