
Chemistry & Chemical Reactivity
10th Edition
ISBN: 9781337399074
Author: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 7, Problem 71SCQ
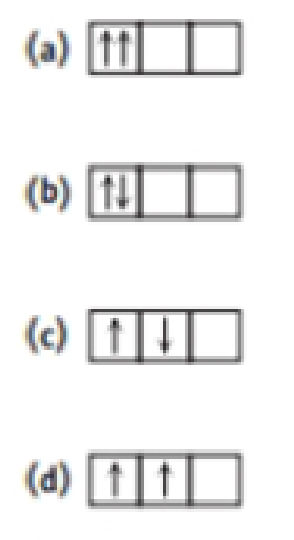
Compare the configurations below with two electrons located in p orbitals. Which would be the most stable (have the lowest energy)? Which would be the least stable? Explain your answers.
Expert Solution & Answer
Trending nowThis is a popular solution!

Chapter 7 Solutions
Chemistry & Chemical Reactivity
Ch. 7.3 - (a) What element has the configuration...Ch. 7.3 - Write one possible set of quantum numbers for the...Ch. 7.3 - Using the periodic table and without looking at...Ch. 7.4 - Prob. 7.4CYUCh. 7.5 - Without looking at the figures for the periodic...Ch. 7.6 - The most common oxidation state of a rare earth...Ch. 7.6 - Prob. 1.2ACPCh. 7.6 - Prob. 1.3ACPCh. 7.6 - Use the atomic radii of scandium, yttrium,...Ch. 7.6 - Prob. 1.5ACP
Ch. 7.6 - Prob. 1.6ACPCh. 7.6 - Give the electron configurations for iron and the...Ch. 7.6 - Prob. 2.2ACPCh. 7.6 - Prob. 2.3ACPCh. 7.6 - Prob. 2.4ACPCh. 7 - Write the electron configurations for P and CI...Ch. 7 - Write the electron configurations for Mg and Ar...Ch. 7 - Using spdf notation, write the electron...Ch. 7 - Using spdf notation, give the electron...Ch. 7 - Prob. 5PSCh. 7 - Prob. 6PSCh. 7 - Use noble gas and spdf notations to depict...Ch. 7 - The lanthanides, once called the rare earth...Ch. 7 - Prob. 9PSCh. 7 - Prob. 10PSCh. 7 - What is the maximum number of electrons that can...Ch. 7 - What is the maximum number of electrons that can...Ch. 7 - Depict the electron configuration for magnesium...Ch. 7 - Depict the electron configuration for phosphorus...Ch. 7 - Using an orbital box diagram and noble gas...Ch. 7 - Using an orbital box diagram and noble gas...Ch. 7 - Prob. 17PSCh. 7 - Which of the following statements correctly...Ch. 7 - Prob. 19PSCh. 7 - Prob. 20PSCh. 7 - Using orbital box diagrams, depict an electron...Ch. 7 - Prob. 22PSCh. 7 - Prob. 23PSCh. 7 - Using orbital box diagrams and noble gas notation,...Ch. 7 - Manganese is found as MnO2 in deep ocean deposits....Ch. 7 - One compound found in alkaline batteries is NiOOH,...Ch. 7 - Prob. 27PSCh. 7 - Arrange the following elements in order of...Ch. 7 - Prob. 29PSCh. 7 - Prob. 30PSCh. 7 - Which of the following groups of elements is...Ch. 7 - Arrange the following atoms in order of increasing...Ch. 7 - Compare the elements Na, Mg, O, and P. (a) Which...Ch. 7 - Compare the elements B. Al, C, and Si. (a) Which...Ch. 7 - Explain each answer briefly. (a) Place the...Ch. 7 - Explain each answer briefly. (a) Rank the...Ch. 7 - Identify the element that corresponds to each of...Ch. 7 - Identify the element that corresponds to each of...Ch. 7 - Explain why the photoelectron spectra of hydrogen...Ch. 7 - Sketch the major features (number of peaks and...Ch. 7 - These questions are not designated as to type or...Ch. 7 - The deep blue color of sapphires comes from the...Ch. 7 - Using an orbital box diagram and noble gas...Ch. 7 - Prob. 44GQCh. 7 - Prob. 45GQCh. 7 - Prob. 46GQCh. 7 - Which of the following is not an allowable set of...Ch. 7 - A possible excited state for the H atom has an...Ch. 7 - The magnet in the following photo is made from...Ch. 7 - Name the element corresponding to each...Ch. 7 - Arrange the following atoms in order of increasing...Ch. 7 - Prob. 52GQCh. 7 - Answer the questions below about the elements A...Ch. 7 - Answer (he following questions about the elements...Ch. 7 - Which of the following ions are unlikely to be...Ch. 7 - Prob. 56GQCh. 7 - Answer each of the following questions: (a) Of the...Ch. 7 - Prob. 58GQCh. 7 - Prob. 59GQCh. 7 - Two elements in the second transition series (Y...Ch. 7 - Prob. 61GQCh. 7 - The configuration of an element is given here. (a)...Ch. 7 - Answer the questions below about the elements A...Ch. 7 - Answer the questions below concerning ground state...Ch. 7 - Nickel(II) formate [Ni(HCO2)2] is widely used as a...Ch. 7 - Spinets are solids with the general formula M2+...Ch. 7 - The following questions use concepts from this and...Ch. 7 - Which ions in the following list are not likely to...Ch. 7 - Answer the following questions about first...Ch. 7 - The ionization of the hydrogen atom can be...Ch. 7 - Compare the configurations below with two...Ch. 7 - Prob. 72SCQCh. 7 - Write electron configurations to show the first...Ch. 7 - Prob. 74SCQCh. 7 - (a) Explain why the sizes of atoms change when...Ch. 7 - Which of the following elements has the greatest...Ch. 7 - Prob. 77SCQCh. 7 - Prob. 78SCQCh. 7 - The energies of the orbitals in many elements have...Ch. 7 - The ionization energies for the removal of the...Ch. 7 - Using your knowledge of the trends in element...Ch. 7 - Prob. 82SCQCh. 7 - Prob. 83SCQCh. 7 - Prob. 84SCQCh. 7 - Thionyl chloride. SOCl2, is an important...Ch. 7 - Prob. 86SCQCh. 7 - Slaters rules are a way to estimate the effective...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
Recommended textbooks for you
 Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning


Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning
Quantum Numbers, Atomic Orbitals, and Electron Configurations; Author: Professor Dave Explains;https://www.youtube.com/watch?v=Aoi4j8es4gQ;License: Standard YouTube License, CC-BY
QUANTUM MECHANICAL MODEL/Atomic Structure-21E; Author: H to O Chemistry;https://www.youtube.com/watch?v=mYHNUy5hPQE;License: Standard YouTube License, CC-BY